Overview
The Intellis™ with AdaptiveStim™ implantable neurostimulator is powered by proprietary Overdrive™ battery technology. It is designed to overcome limitations with other SCS systems and is optimized for the increased energy demands of High Dose (HD) therapy. Medtronic exclusive features on the Intellis™ platform include AdaptiveStim™ , wireless trialing system, and SureScan™ full-body MRI technologies.
Indications
A Medtronic implantable neurostimulation system is indicated for spinal cord stimulation (SCS) as an aid in the management of chronic, intractable pain of the trunk and/or limbs – including unilateral or bilateral pain associated with the following
conditions:
Failed Back Syndrome (FBS) or low back syndrome or failed
back
Radicular pain syndrome or radiculopathies resulting in pain
secondary to FBS or herniated disk
Postlaminectomy pain
Multiple back operations
Unsuccessful disk surgery
Degenerative Disk Disease (DDD)/herniated disk pain
refractory to conservative and surgical interventions
Peripheral causalgia
Epidural fibrosis
Arachnoiditis or lumbar adhesive arachnoiditis
Complex Regional Pain Syndrome (CRPS), Reflex Sympathetic Dystrophy (RSD), or causalgia
Product Details
Intellis™ with AdaptiveStim™ Implantable Neurostimulator
Smallest fully implantable spinal cord neurostimulator
~1 hour recharge — empty to full (approximate time)
Improved battery performance with minimal capacity fade (<5%)
Designed for patient comfort and more flexible placement during the implant
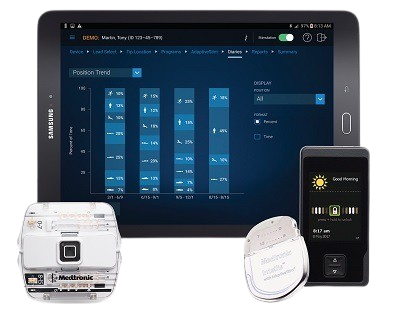
AdaptiveStim™ technology automatically adjusts the therapy as your patient moves, delivering a personalized treatment based on seven unique body positions.
Snapshot™ reporting, powered by AdaptiveStim™ technology, allows physicians to objectively monitor a patient's progress.

| Model number | 97715 |
| Weight | 29.1g (1.0oz) |
| Height | 57.1mm (2.2in) |
| Width | 47.2mm (1.9in) |
| Battery Type | Rechargeable |
| Volume | 13.9 cc |
Intellis™ LT
Smallest fully implantable spinal cord neurostimulator
~1 hour recharge — empty to full (approximate time)
Improved battery performance with minimal capacity fade (<5%)
Designed for patient comfort and more flexible placement during the implant

| Model number | 97716 |
| Weight | 29.1g (1.0oz) |
| Height | 57.1mm (2.2in) |
| Width | 47.2mm (1.9in) |
| Battery Type | Rechargeable |
| Volume | 13.9 cc |
Intellis Platform Features
Wireless External Neurostimulator
Wireless, sterile single-use external neurostimulator simplifies testing during trial and implant
Designed for patient comfort and convenience

| Model number | 97725 |
| Weight | 71g (2.5oz) |
| Length | 79mm (3.1in) |
| Width | 74mm (2.9in) |
| Battery Type | Non-rechargeable |
| Volume | 20mm (0.8in) |
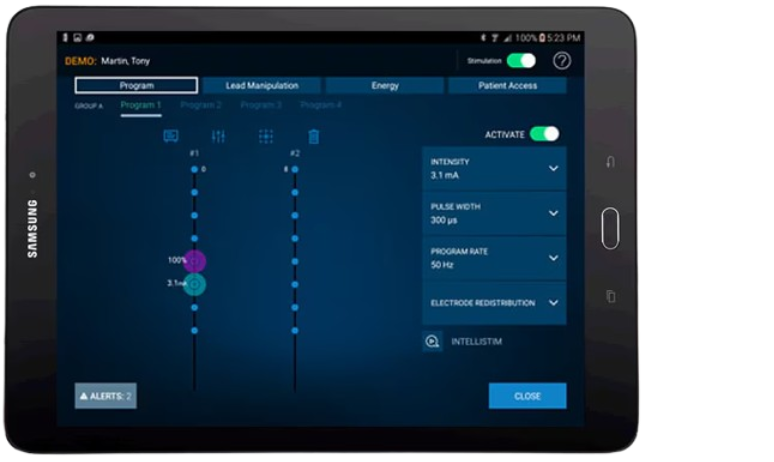
Clinician Programmer
Tablet with touchscreen interface
Secure, wireless programming outside the sterile field
Tailored programming workflows
Electronic transfer of a variety of reports
Bluetooth® communication
Requires the use of the Intellis™ clinician programmer app and communicator device
OVERDRIVE™ BATTERY
Unrivaled battery chemistry performance
>95% battery capacity retained at 9 years, independent of therapy parameters or recharge preferences
Optimized for the increased energy demands of HD therapies.
Up to 3X faster recharge than traditional lithium ion batteries
About 1-hour recharge from empty to full
Ability to recharge a completely discharged device

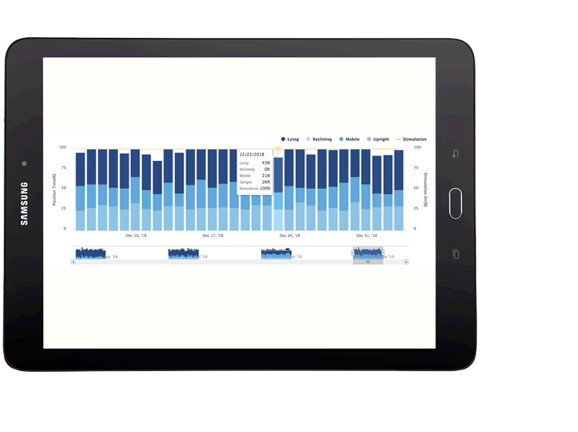
SNAPSHOT™ REPORTING
Enabled by AdaptiveStim™ Technology†
Records patient outcomes, including functional activity
Offers view of stimulation usage, position, and recharge trends
Enables 24/7 secure access, sharing, and storage via cloud-based platform1
Combined Patient Programmer and Recharger
Recharge from fully depleted battery
Easy-to-adjust therapy
Easy-to-read interface
Large, pliable recharge antenna designed for patient comfort

